Less meat, more plants: top US heart body’s diet advice at odds with MahaAmerican Heart Association bucks Trump administration line to suggest prioritizing plant-based protein over meat In contrast to the federal government’s recommendation of use of beef tallow and consumption of red meat, the AHA pushed for plant-based proteins such as legumes, nuts and seeds. Photograph: Allen J Schaben/Los Angeles Times/Getty Images The American Heart Association’s new nutrition guidance, released on Tuesday, emphasizes a dietary pattern rich in vegetables, fruits and whole grains, prioritizing plant-based protein over meat. It also suggests limiting the use of sugar, salt and ultra-processed foods and replacing full fat dairy with non-fat and low-fat dairy. Donald Trump earlier this year announced a new slate of dietary guidelines that recommended Americans eat more protein from both animal and plant sources, and encouraged the consumption of full-fat dairy. In contrast to the federal government’s recommendation of use of beef tallow and consumption of red meat, the AHA is pushing for plant-based proteins such as legumes, nuts and seeds, choosing low-fat or fat-free dairy, and, when consuming red meat, selecting lean cuts, avoiding processed forms and keeping portions small. The Trump administration and health secretary Robert F Kennedy Jr’s push to “Make America healthy again” has focused on the removal of synthetic dyes, other chemical ingredients, ultra-processed foods, consumption of less sugar and seed oils. The AHA also suggests Americans choose sources of unsaturated fats, avoid ultraprocessed foods and minimize intake of added sugars in beverages and foods. The organization recommended Americans to prepare foods with minimal or no salt, and limit the intake of alcohol. The guidance also recommended children can and should begin following a heart-healthy dietary pattern starting at one year old. The AHA’s guidance was aligned with the US Food and Drug Administration’s dietary guidelines on major issues, a spokesperson from the FDA said, adding they looked forward to working collaboratively with AHA. The AHA, which is the nation’s oldest and largest voluntary organization dedicated to fighting heart disease and stroke, releases dietary guidance about every five years to promote cardiovascular health. ARTICLE SOURCE: https://www.theguardian.com/us-news/2026/apr/01/american-heart-association-dietary-guidelines-maha









FDA warns consumers not to use counterfeit Ozempic (semaglutide) found in U.S. drug supply chain
[12/21/2023] FDA continues to investigate counterfeit Ozempic (semaglutide) injection 1 milligram (mg) in the legitimate U.S. drug supply chain and has seized thousands of units of the product. The agency advises wholesalers, retail pharmacies, health care practitioners and patients to check the product they have received and not distribute, use, or sell products labeled with lot number NAR0074 and serial number 430834149057 as pictured below. Some counterfeit products may still be available for purchase.
FDA and Novo Nordisk (manufacturer of Ozempic) are testing the seized products and do not yet have information about the drugs’ identity, quality, or safety.
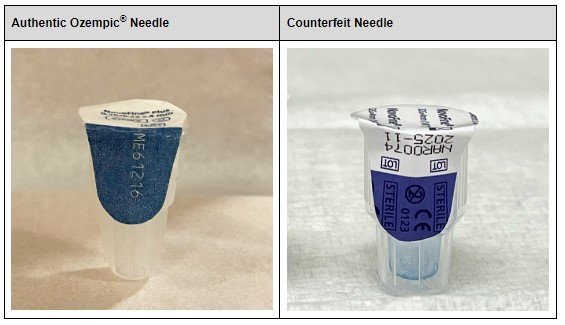
Additionally, analysis found the needles from the samples are counterfeit. Accordingly, the sterility of the needles cannot be confirmed, which presents an increased risk of infection for patients who use the counterfeit products. Based on analyses completed to date, other confirmed counterfeit components within the seized products are the pen label, accompanying health care professional and patient information, and carton.
FDA is aware of five adverse events from this lot, none of which are serious and are consistent with known common adverse reactions to authentic Ozempic, which are nausea, vomiting, diarrhea, abdominal pain and constipation.
FDA recommends retail pharmacies only purchase authentic Ozempic through authorized distributors of Novo NordiskExternal Link Disclaimer and review the photographs and information to confirm the legitimacy of their shipments. Patients should only obtain Ozempic with a valid prescription through state-licensed pharmacies and check the product before using for any signs of counterfeiting.
FDA takes reports of possible counterfeit products seriously and works closely with other federal agencies and the private sector to help protect the nation’s drug supply. FDA’s investigation is ongoing, and the agency is working with Novo Nordisk to identify, investigate, and remove further suspected counterfeit semaglutide injectable products found in the U.S.
Health care professionals and consumers should report adverse events or side effects related to the use of this product to FDA’s MedWatch Safety Information and Adverse Event Reporting Program:
Entities, including online sellers, selling counterfeit and/or tampered medicines should be reported to FDA. Suspected counterfeit products may be reported to FDA by calling your local FDA consumer complaint coordinator or by reporting it directly at report suspected criminal activity.
Retailers and patients may also contact Novo Nordisk customer care at 1-800-727-6500 Monday through Friday from 8:30 a.m. to 6 p.m. ET with questions or concerns.
Visuals of authentic and counterfeit needles are shown below:
More Information:
Medications Containing Semaglutide Marketed for Type 2 Diabetes or Weight Loss
ARTICLE SOURCE:
https://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-consumers-not-use-counterfeit-ozempic-semaglutide-found-us-drug-supply-chain
Subscribe to Carnivore Talk on YouTube | Be our guest on the channel | Leave me a voicemail, yo!